СТСАЕ , Common Terminology Criteria for Adverse Events , ECOG, Eastern Cooperative Oncology Group, NYHA, New York Heart Association, PD-L1, лиганд 1 белка программируемой клеточной гибели, Pt, препараты платины, СКФ, скорость клубочковой фильтрации, СН, сердечная недостаточность, ХТ, химиотерапия
1. Oken MM, Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982 Dec;5(6):649-655.
2. Common Terminology Criteria for Adverse Events (CTCAE) v5.0 https://ctep.cancer.gov/protocoldevelopment/ electronic_applications/ctc.htm
3. Jiang DM, Gupta S, Kitchlu A, et al. Defining cisplatin eligibility in patients with muscle-invasive bladder cancer. Nat Rev Urol 2021; 18: 104–114.
4. Delgado C, A Unifying Approach for GFR Estimation: Recommendations of the NKF-ASN Task Force on Reassessing the Inclusion of Race in Diagnosing Kidney Disease. Am J Kidney Dis. 2022 Feb;79(2):268-288.e1.
5. Stecca C, Abdeljalil O, Sridhar SS. Metastatic Urothelial Cancer: a rapidly changing treatment landscape. Ther Adv Med Oncol. 2021 Sep 30;13:17588359211047352.
6. Powles T, et al., Bladder cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022 Mar;33(3):244-258;
7. NCCN Guidelines: Bladder Cancer,. www.nccn.org
1. Oken MM, Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982 Dec;5(6):649-655.
2. Common Terminology Criteria for Adverse Events (CTCAE) v5.0 https://ctep.cancer.gov/protocoldevelopment/ electronic_applications/ctc.htm
3. Jiang DM, Gupta S, Kitchlu A, et al. Defining cisplatin eligibility in patients with muscle-invasive bladder cancer. Nat Rev Urol 2021; 18: 104–114.
4. Delgado C, A Unifying Approach for GFR Estimation: Recommendations of the NKF-ASN Task Force on Reassessing the Inclusion of Race in Diagnosing Kidney Disease. Am J Kidney Dis. 2022 Feb;79(2):268-288.e1.
5. Stecca C, Abdeljalil O, Sridhar SS. Metastatic Urothelial Cancer: a rapidly changing treatment landscape. Ther Adv Med Oncol. 2021 Sep 30;13:17588359211047352.
6. Powles T, et al., Bladder cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022 Mar;33(3):244-258;
7. NCCN Guidelines: Bladder Cancer,. www.nccn.org
Местнораспространенный неоперабельный или метастатический УР
АЛГОРИТМ ОПРЕДЕЛЕНИЯ ПЕРЕНОСИМОСТИ ПРЕПАРАТОВ ПЛАТИНЫ
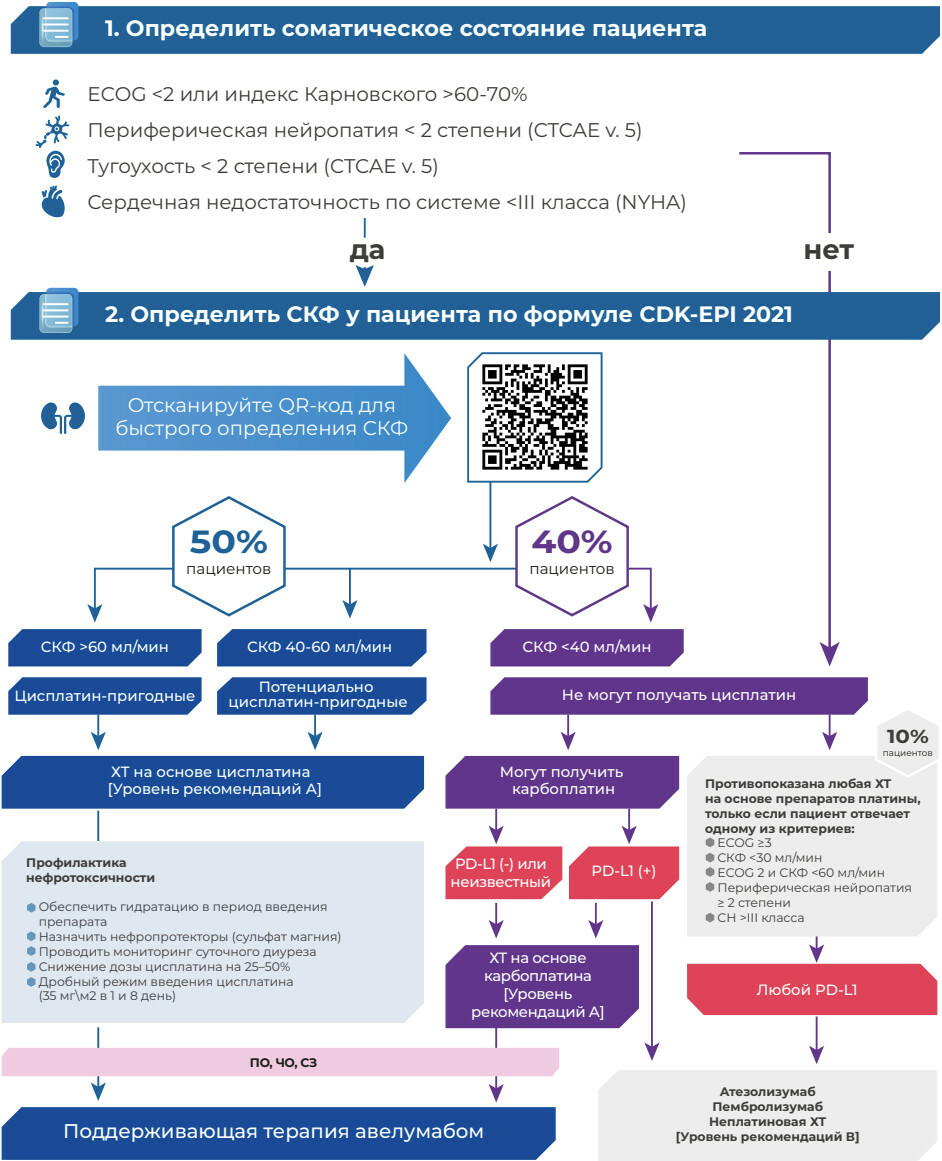
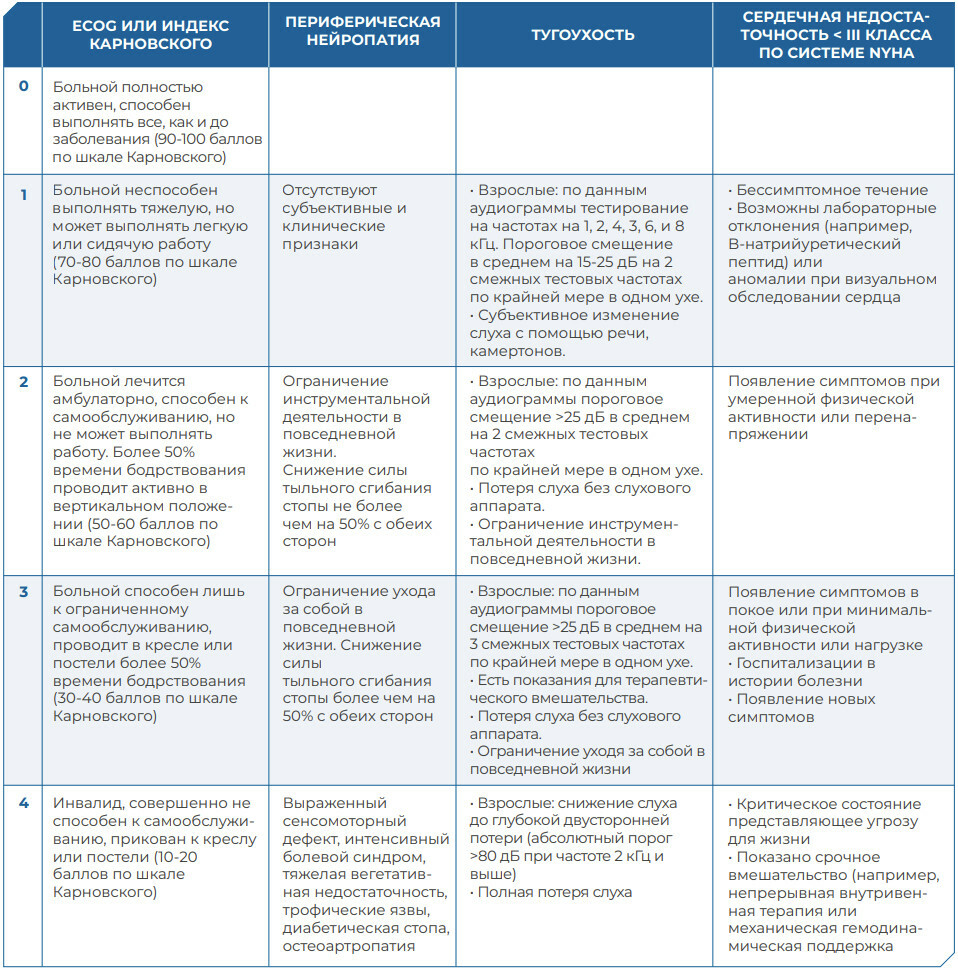
АЛГОРИТМ ОПРЕДЕЛЕНИЯ ПЕРЕНОСИМОСТИ ПРЕПАРАТОВ ПЛАТИНЫ

RU-AVE-00284 | Сентябрь 2023 Чтобы ознакомиться с инструкцией по медицинскому применению препарата Бавенсио® (авелумаб), отсканируйте QR-код или перейдите по ссылке. РУ: ЛП-005866. © ООО «Мерк» 115054, Москва, ул. Валовая, д. 35, БЦ “Wall Street”, этаж 6 Тел. +7(495) 937-33-05 Веб-сайт: www.merckgroup.com/ru-ru Почта: russia@merckgroup.com Официальный сайт Мерк Онкология в России https://merck.oncology.ru/